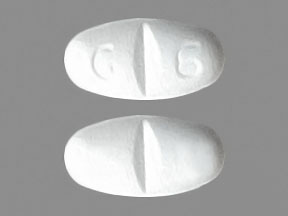
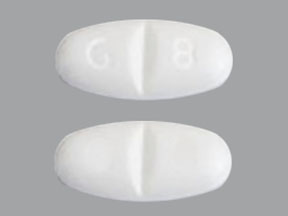
Pill with imprint NT 16 is White, Elliptical/Oval and has been identified as Gabapentin 600 mg. It is supplied by Greenstone LLC.
Gabapentin is used in the treatment of postherpetic neuralgia; epilepsy and belongs to the drug class gamma-aminobutyric acid analogs. Risk cannot be ruled out during pregnancy. Gabapentin 600 mg is not a controlled substance under the Controlled Substances Act (CSA).
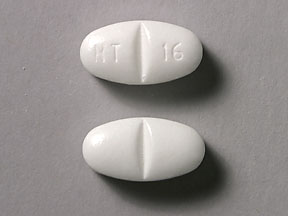
Gabapentin
- Imprint
- NT 16
- Strength
- 600 mg
- Color
- White
- Shape
- Elliptical/Oval
- Availability
- Prescription only
- Drug Class
- Gamma-aminobutyric acid analogs
- Pregnancy Category
- C – Risk cannot be ruled out
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Greenstone LLC
- National Drug Code (NDC)
- 59762-4355
-
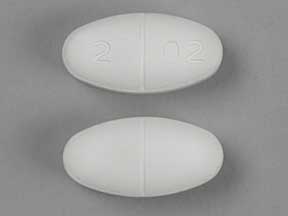
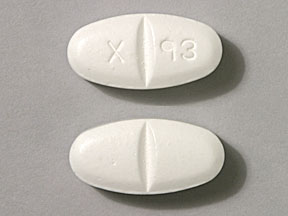
NT 26 (Gabapentin 800 mg)
Pill with imprint NT 26 is White, Elliptical/Oval and has been identified as Gabapentin 800 mg. It is supplied by Greenstone LLC.
Gabapentin is used in the treatment of postherpetic neuralgia; epilepsy and belongs to the drug class gamma-aminobutyric acid analogs. Risk cannot be ruled out during pregnancy. Gabapentin 800 mg is not a controlled substance under the Controlled Substances Act (CSA).
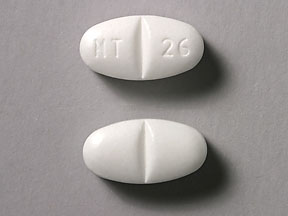
Gabapentin
- Imprint
- NT 26
- Strength
- 800 mg
- Color
- White
- Shape
- Elliptical/Oval
- Availability
- Prescription only
- Drug Class
- Gamma-aminobutyric acid analogs
- Pregnancy Category
- C – Risk cannot be ruled out
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Greenstone LLC
- National Drug Code (NDC)
- 59762-4460